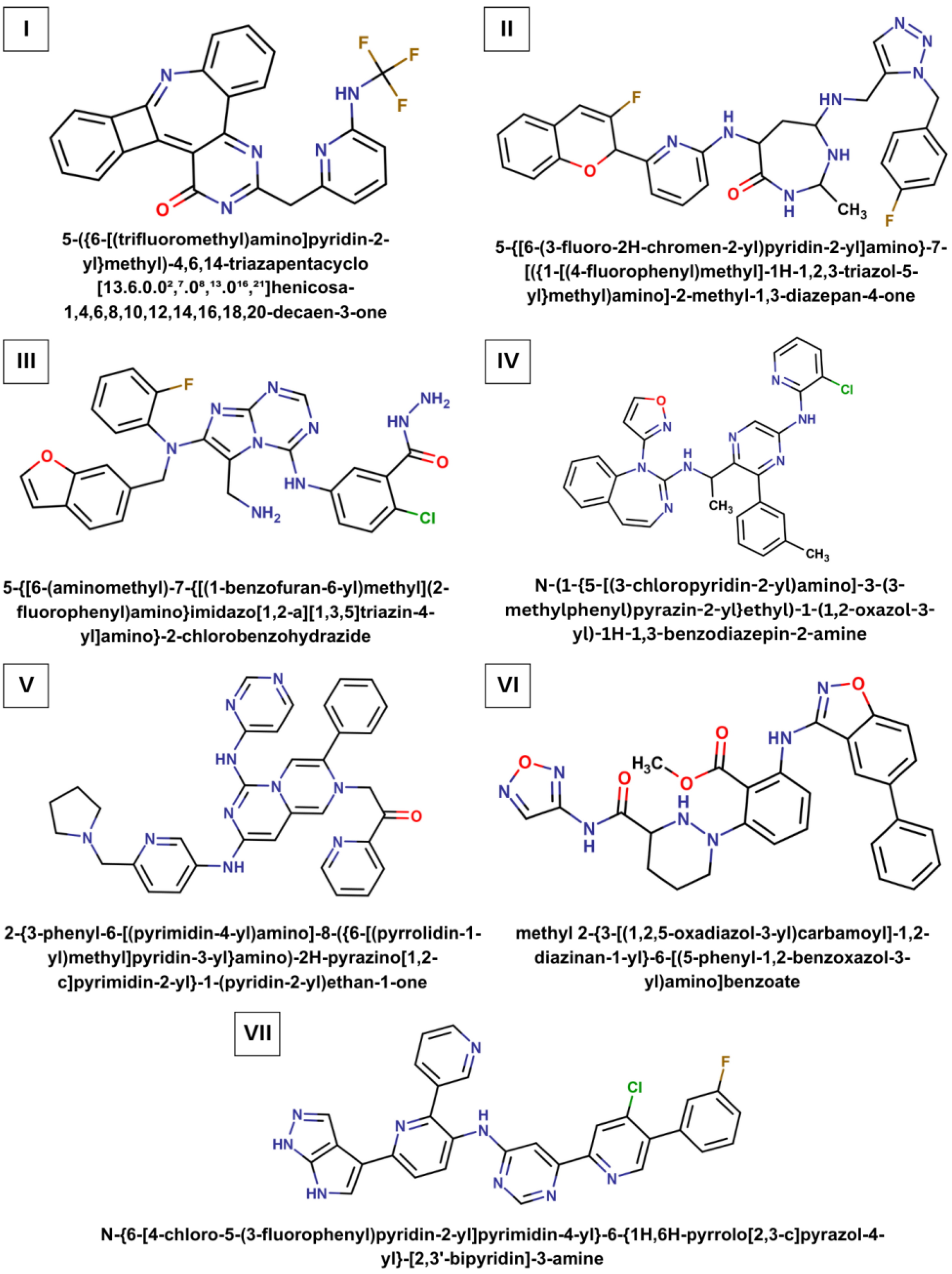
IJMS | Free Full-Text | AI-Driven De Novo Design and Molecular Modeling for Discovery of Small-Molecule Compounds as Potential Drug Candidates Targeting SARS-CoV-2 Main Protease

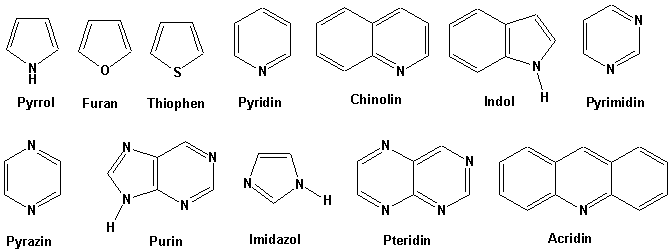
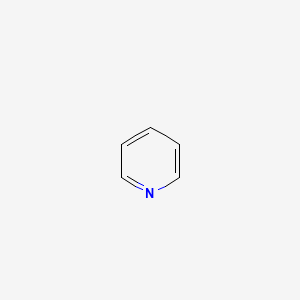
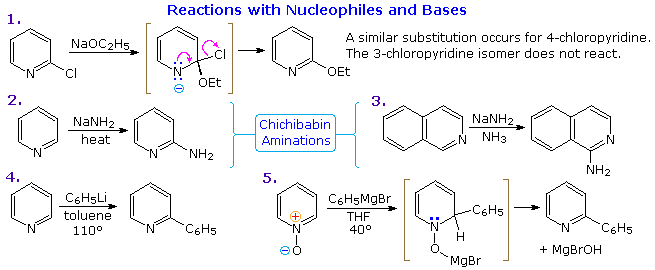
Metallation of pyridin-2-yldiazines. Use of pyridine ring as ortho-directing group. Diazines. Part 45 - ScienceDirect

DE102009016374A1 - Preparing a 2-amino pyridine derivative comprises reacting an open-chain nitrile precursor with a nitrogen containing compound in a cyclization reaction - Google Patents

Oxyfunctionalization of pyridine derivatives using whole cells of Burkholderia sp. MAK1 | Scientific Reports
![tert-butyl N-[5-(hydroxymethyl)pyridin-2-yl]carbamate_Pyridines_Hetero 6-membered Rings +_By Structure_Diversity Oriented Building Blocks_affinitychem tert-butyl N-[5-(hydroxymethyl)pyridin-2-yl]carbamate_Pyridines_Hetero 6-membered Rings +_By Structure_Diversity Oriented Building Blocks_affinitychem](http://affinitychem.com/images/201806/source_img/10634_G_1528158572284.jpg)
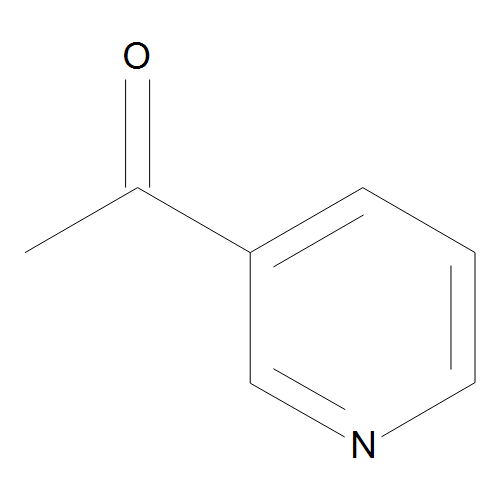
tert-butyl N-[5-(hydroxymethyl)pyridin-2-yl]carbamate_Pyridines_Hetero 6-membered Rings +_By Structure_Diversity Oriented Building Blocks_affinitychem

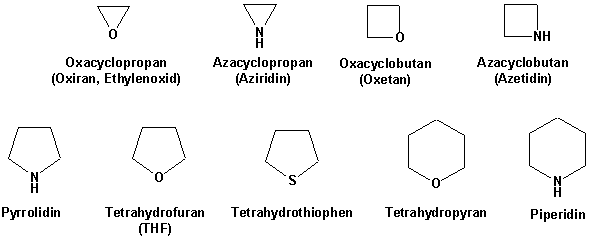
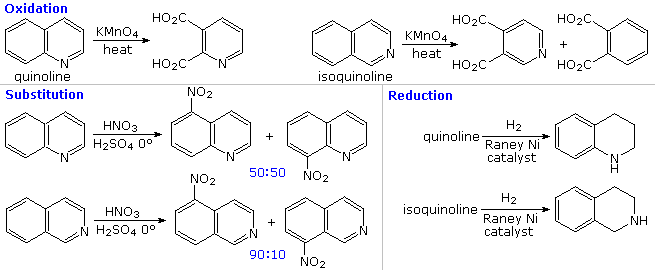
Polarization of the Pyridine Ring: Highly Functionalized Piperidines from Tungsten−Pyridine Complex | Journal of the American Chemical Society
![Synthesis of N-Alkylpyridin-4-ones and Thiazolo[3,2-A]pyridin-5-ones Through Pummerer-Type Reactions | Organic Chemistry | ChemRxiv | Cambridge Open Engage Synthesis of N-Alkylpyridin-4-ones and Thiazolo[3,2-A]pyridin-5-ones Through Pummerer-Type Reactions | Organic Chemistry | ChemRxiv | Cambridge Open Engage](https://chemrxiv.org/engage/api-gateway/chemrxiv/assets/orp/resource/item/60c742b0bdbb8971b9a3854c/largeThumb/synthesis-of-n-alkylpyridin-4-ones-and-thiazolo-3-2-a-pyridin-5-ones-through-pummerer-type-reactions.jpg)
Synthesis of N-Alkylpyridin-4-ones and Thiazolo[3,2-A]pyridin-5-ones Through Pummerer-Type Reactions | Organic Chemistry | ChemRxiv | Cambridge Open Engage
![2206825-87-6 | 5-[[4-[3-(Aminocarbonyl)-1,4,5,7-tetrahydro-1-(4-methoxyphenyl)-7-oxo-6H-pyrazolo[3,4-c] pyridin-6-yl]phenyl]amino]pentanoic Acid | Apixaban Open Chain Acid; 5-[[4-[3-(Aminocarbonyl)-1,4,5,7-tetrahydro-1-(4-methoxyphenyl)-7-oxo-6H ... 2206825-87-6 | 5-[[4-[3-(Aminocarbonyl)-1,4,5,7-tetrahydro-1-(4-methoxyphenyl)-7-oxo-6H-pyrazolo[3,4-c] pyridin-6-yl]phenyl]amino]pentanoic Acid | Apixaban Open Chain Acid; 5-[[4-[3-(Aminocarbonyl)-1,4,5,7-tetrahydro-1-(4-methoxyphenyl)-7-oxo-6H ...](https://www.trc-canada.com/prod-img/A875620.png)
2206825-87-6 | 5-[[4-[3-(Aminocarbonyl)-1,4,5,7-tetrahydro-1-(4-methoxyphenyl)-7-oxo-6H-pyrazolo[3,4-c] pyridin-6-yl]phenyl]amino]pentanoic Acid | Apixaban Open Chain Acid; 5-[[4-[3-(Aminocarbonyl)-1,4,5,7-tetrahydro-1-(4-methoxyphenyl)-7-oxo-6H ...

Novel Pyridyl Ring C5 Substituted Analogues of Epibatidine and 3-(1-Methyl-2(S)- pyrrolidinylmethoxy)pyridine (A-84543) as Highly Selective Agents for Neuronal Nicotinic Acetylcholine Receptors Containing β2 Subunits | Journal of Medicinal Chemistry

AMINO-BENZAZOLES AS P2Y1 RECEPTOR INHIBITORS WITH PYRIDINE RING AND HETEROCYCLIC COMPONENTS - diagram, schematic, and image 29

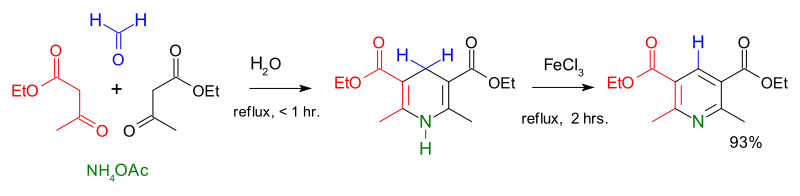
Non-enzymatic pyridine ring formation in the biosynthesis of the rubrolone tropolone alkaloids | Nature Communications
![Optimization of novel benzofuro[3,2-b]pyridin-2(1H)-one derivatives as dual inhibitors of BTK and PI3Kδ - ScienceDirect Optimization of novel benzofuro[3,2-b]pyridin-2(1H)-one derivatives as dual inhibitors of BTK and PI3Kδ - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523418311012-fx1.jpg)