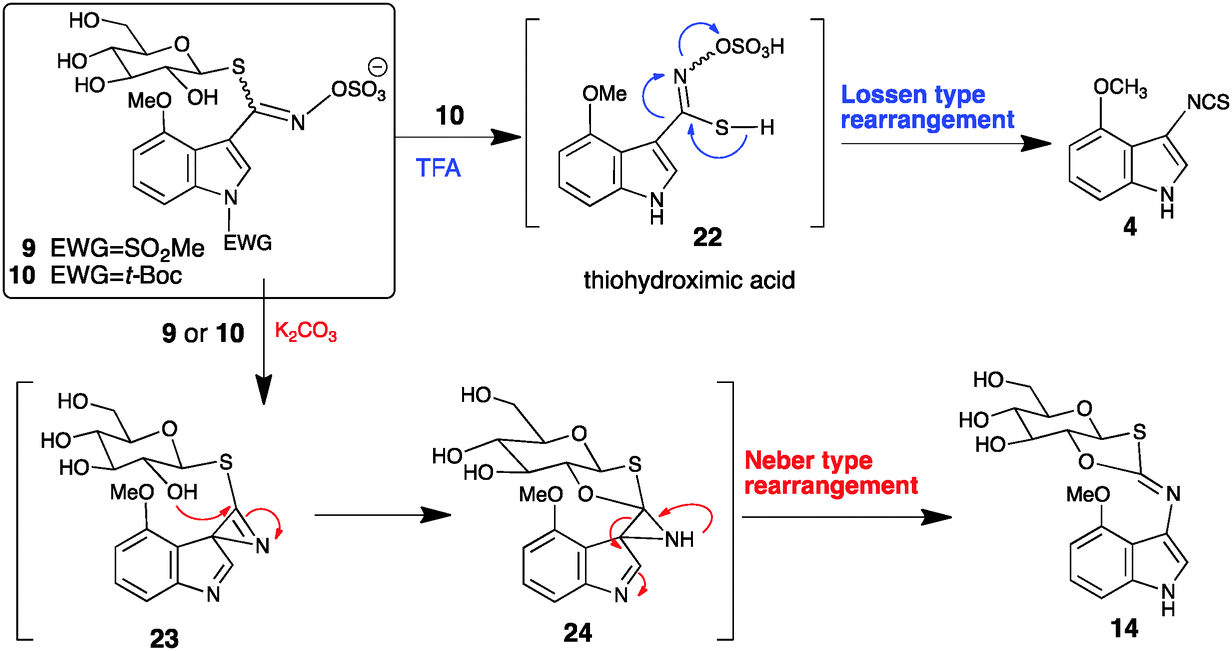
Divergent reactivity of an indole glucosinolate yields Lossen or Neber rearrangement products: the phytoalexin rapalexin A or a unique β- d -glucopyra ... - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC09822J

Synthesis of 2-Unsubstituted Pyrrolidines and Piperidines from Donor–Acceptor Cyclopropanes and Cyclobutanes: 1,3,5-Triazinanes as Surrogates for Formylimines | The Journal of Organic Chemistry

Biomolecules | Free Full-Text | Synthesis and HPLC-ECD Study of Cytostatic Condensed O,N-Heterocycles Obtained from 3-Aminoflavanones | HTML

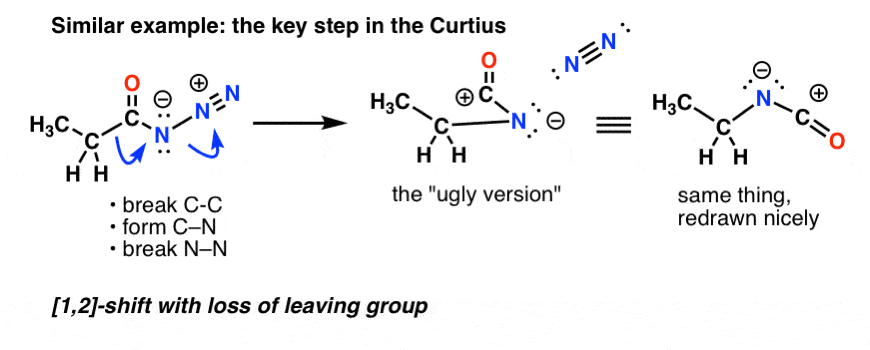
PDF) Organic Reactions Volume 78, Chapter 2, pp. 321-410 The Neber Rearrangement; John Wiley and Sons, 2012
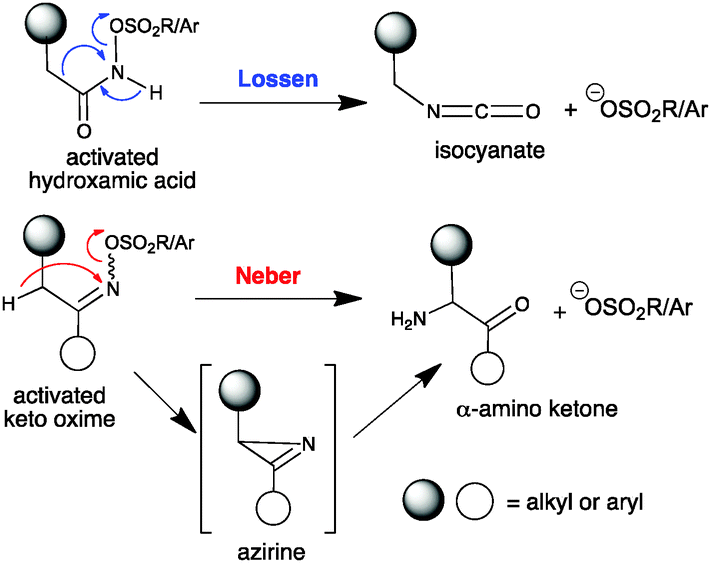
Divergent reactivity of an indole glucosinolate yields Lossen or Neber rearrangement products: the phytoalexin rapalexin A or a unique β- d -glucopyra ... - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC09822J
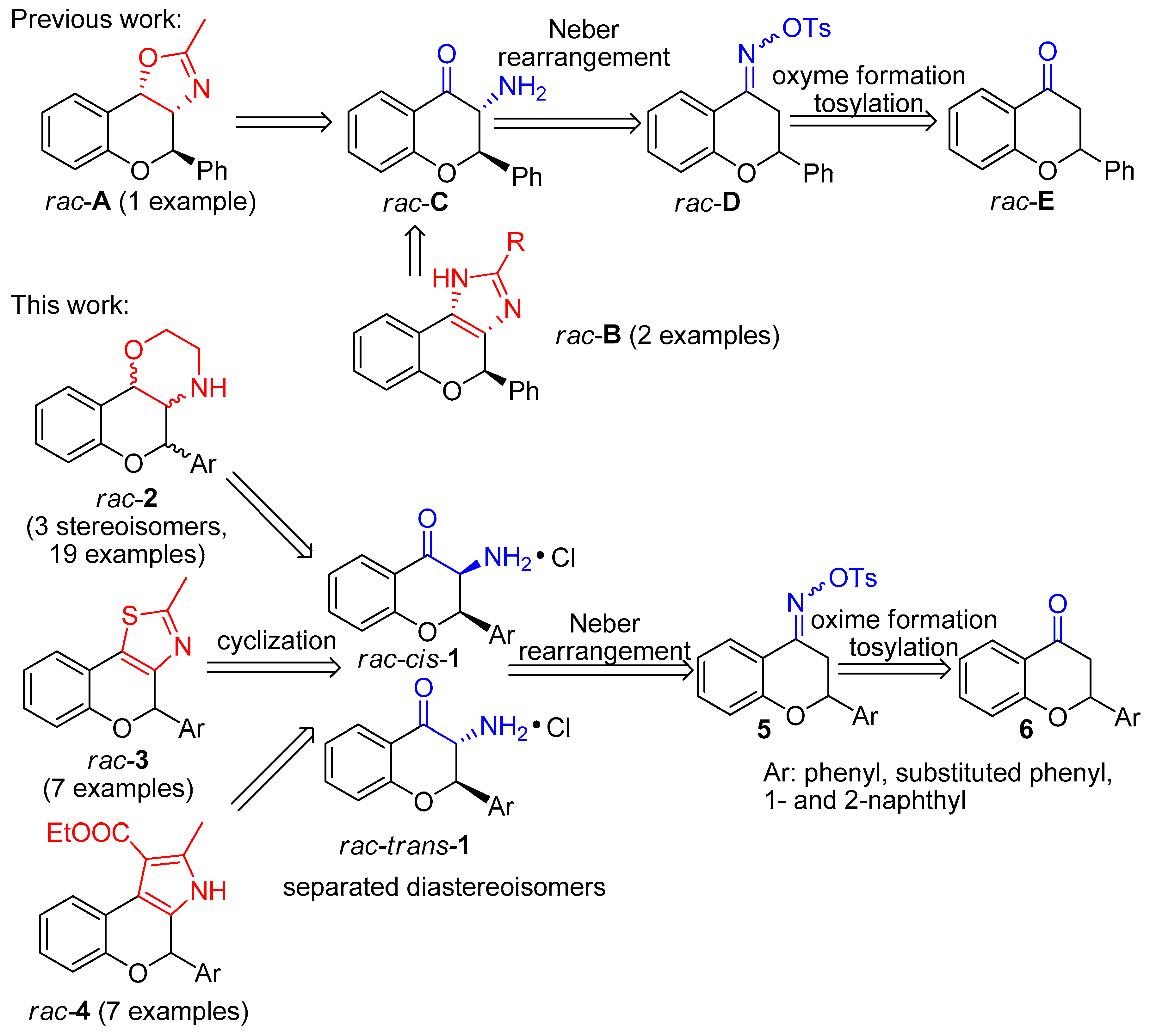
Biomolecules | Free Full-Text | Synthesis and HPLC-ECD Study of Cytostatic Condensed O,N-Heterocycles Obtained from 3-Aminoflavanones | HTML
![AggarwalLab on Twitter: "To mark the start of the Olympics, Kay has provided us with a useful summary on [3,3]-sigmatropic rearrangements: https://t.co/abciN5Gwsz" / Twitter AggarwalLab on Twitter: "To mark the start of the Olympics, Kay has provided us with a useful summary on [3,3]-sigmatropic rearrangements: https://t.co/abciN5Gwsz" / Twitter](https://pbs.twimg.com/media/E29XKq3X0AQyxoF.png)









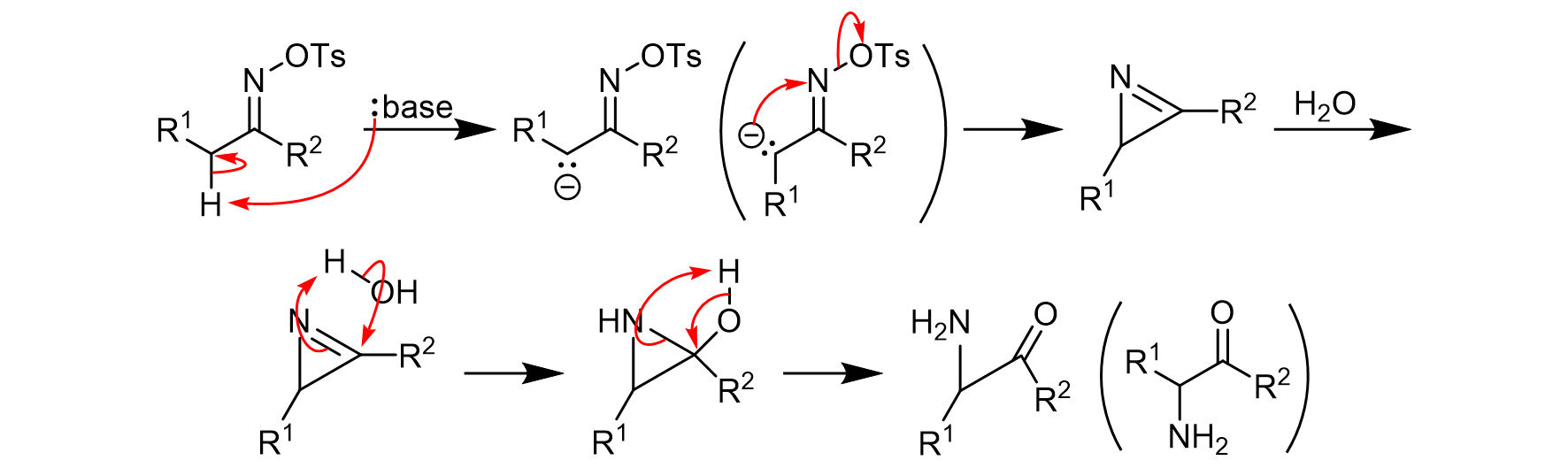

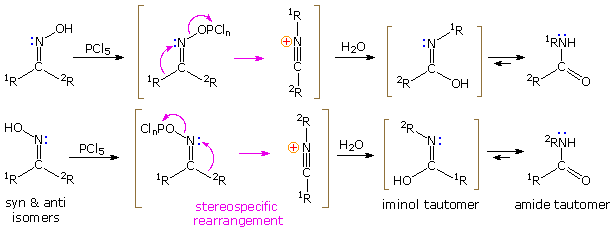






.gif)
