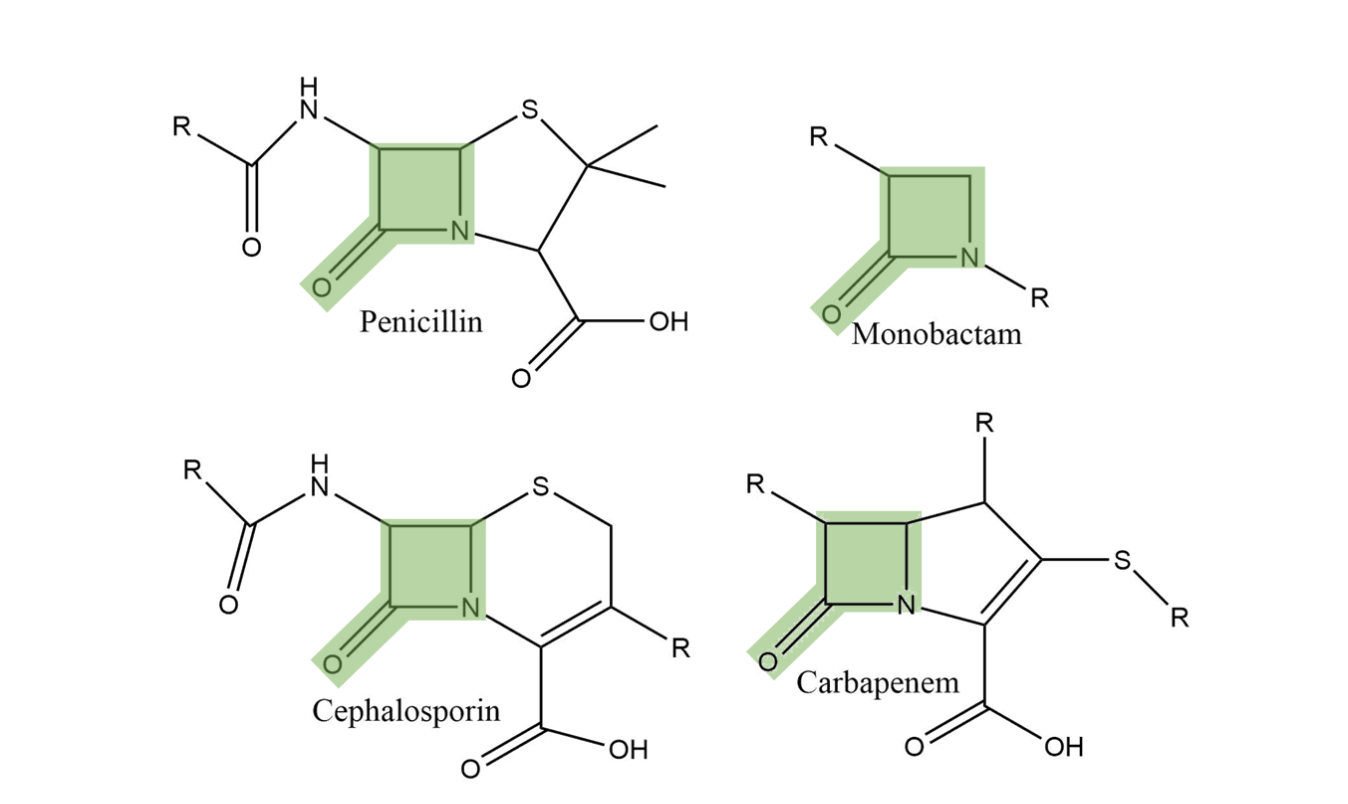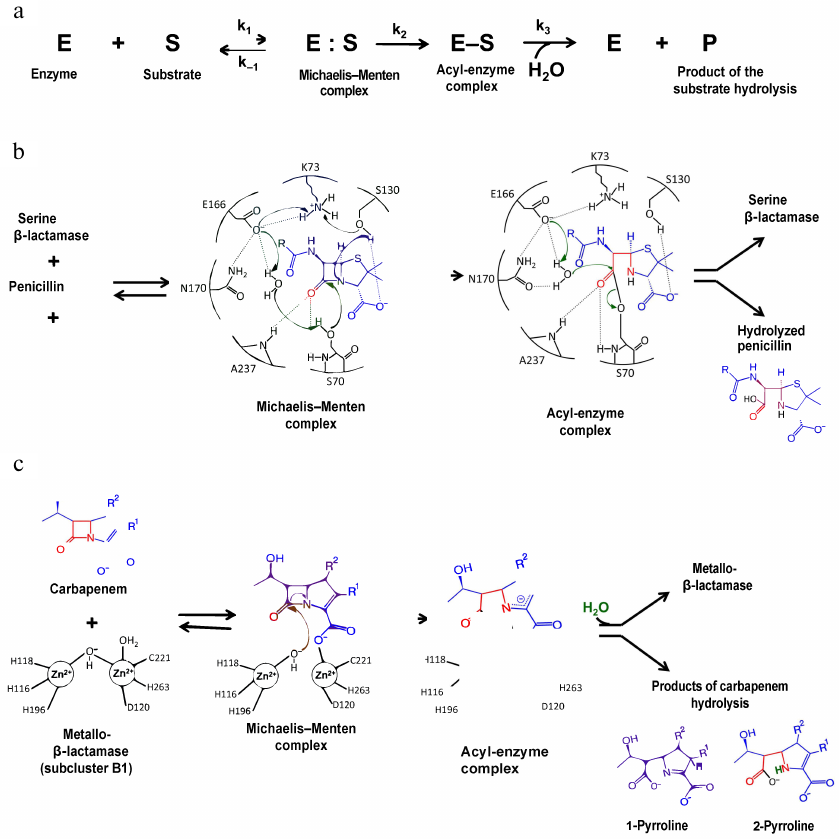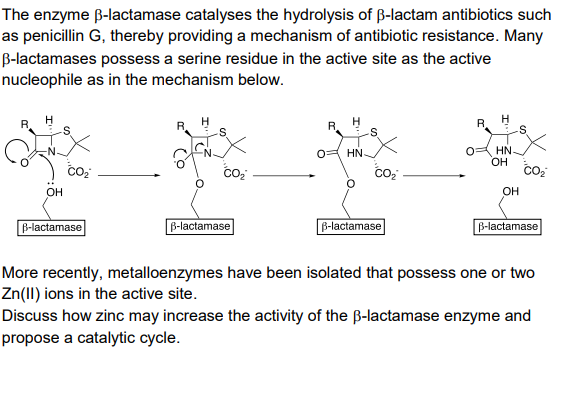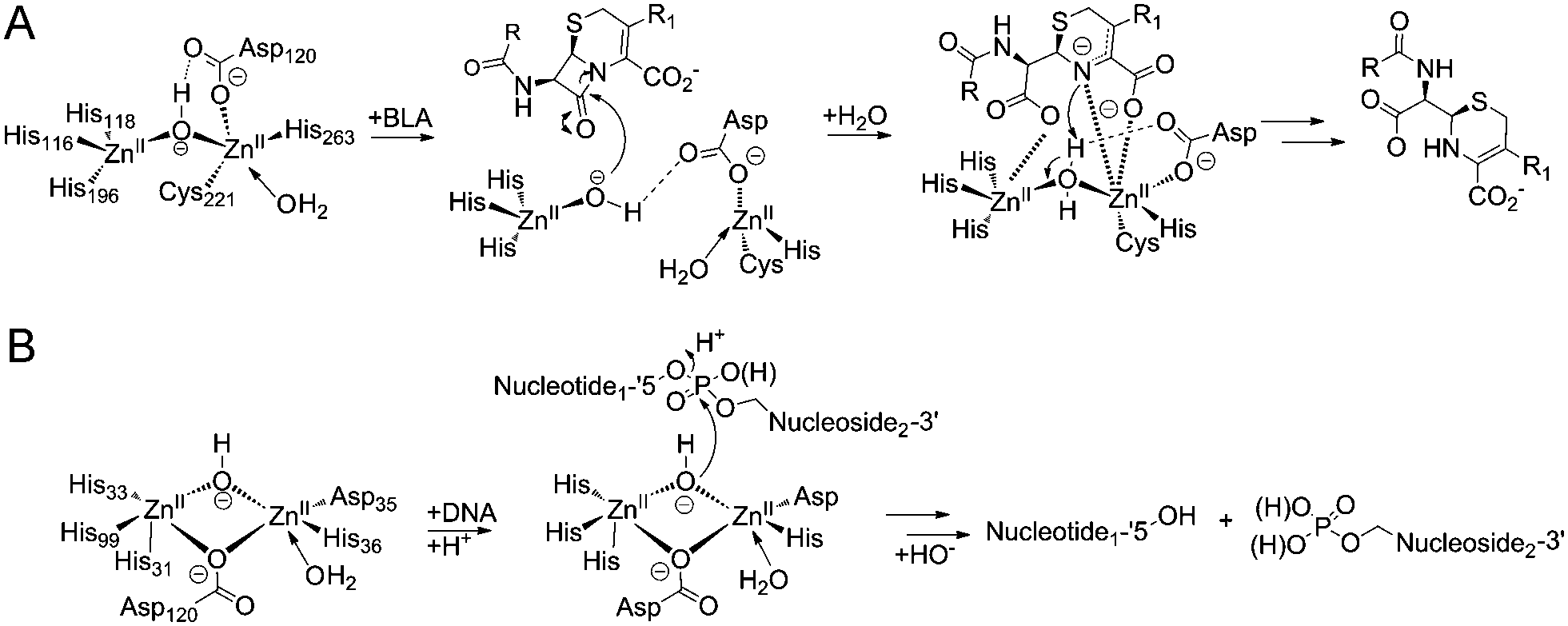
Cephalosporins inhibit human metallo β-lactamase fold DNA repair nucleases SNM1A and SNM1B/apollo - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC00529B
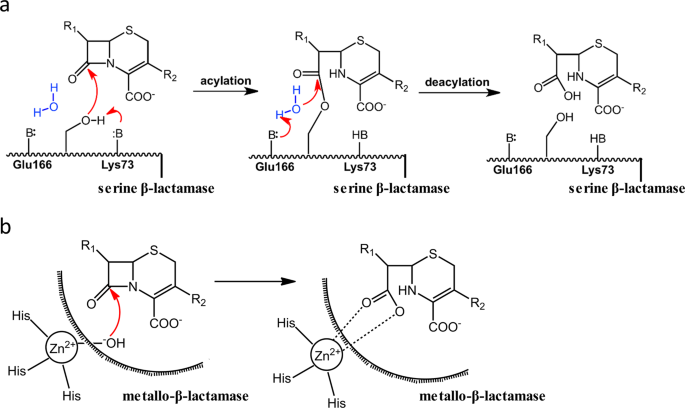
The hydrolytic water molecule of Class A β-lactamase relies on the acyl-enzyme intermediate ES* for proper coordination and catalysis | Scientific Reports

Comparison of mechanisms for hydrolysis of-lactam antibiotics catalyzed... | Download Scientific Diagram

Water | Free Full-Text | Targeted Hydrolysis of β-Lactam Antibiotics in Dry Suspension Residue: A Proposed Method to Reduce Ecological Toxicity and Bacterial Resistance | HTML
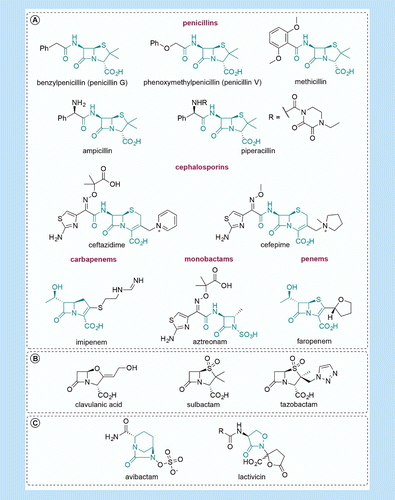
The road to avibactam: the first clinically useful non-β-lactam working somewhat like a β-lactam | Future Medicinal Chemistry
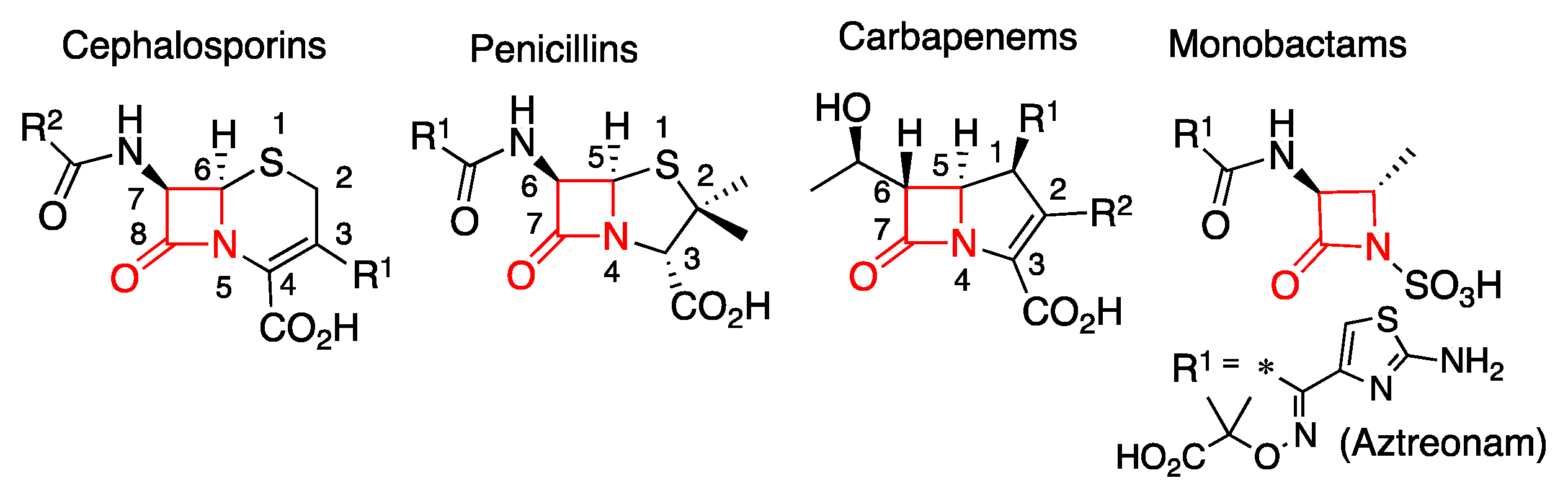
Biomolecules | Free Full-Text | Metallo-β-Lactamase Inhibitors Inspired on Snapshots from the Catalytic Mechanism | HTML
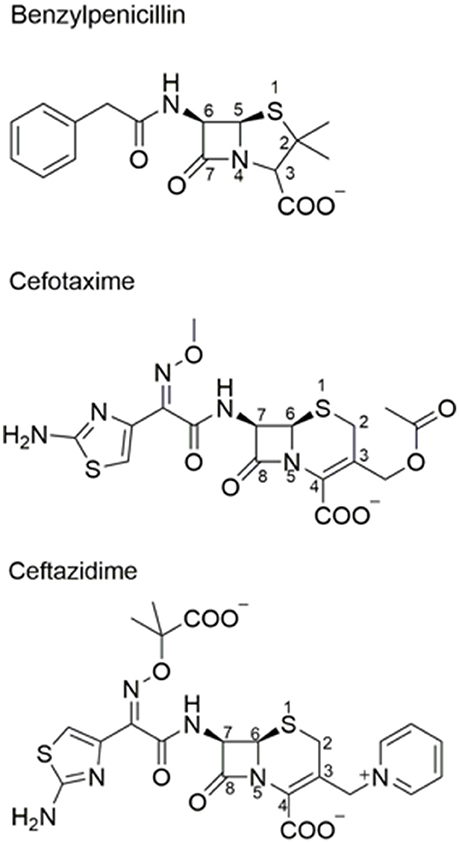
Frontiers | Structural and Mechanistic Basis for Extended-Spectrum Drug-Resistance Mutations in Altering the Specificity of TEM, CTX-M, and KPC β-lactamases | Molecular Biosciences
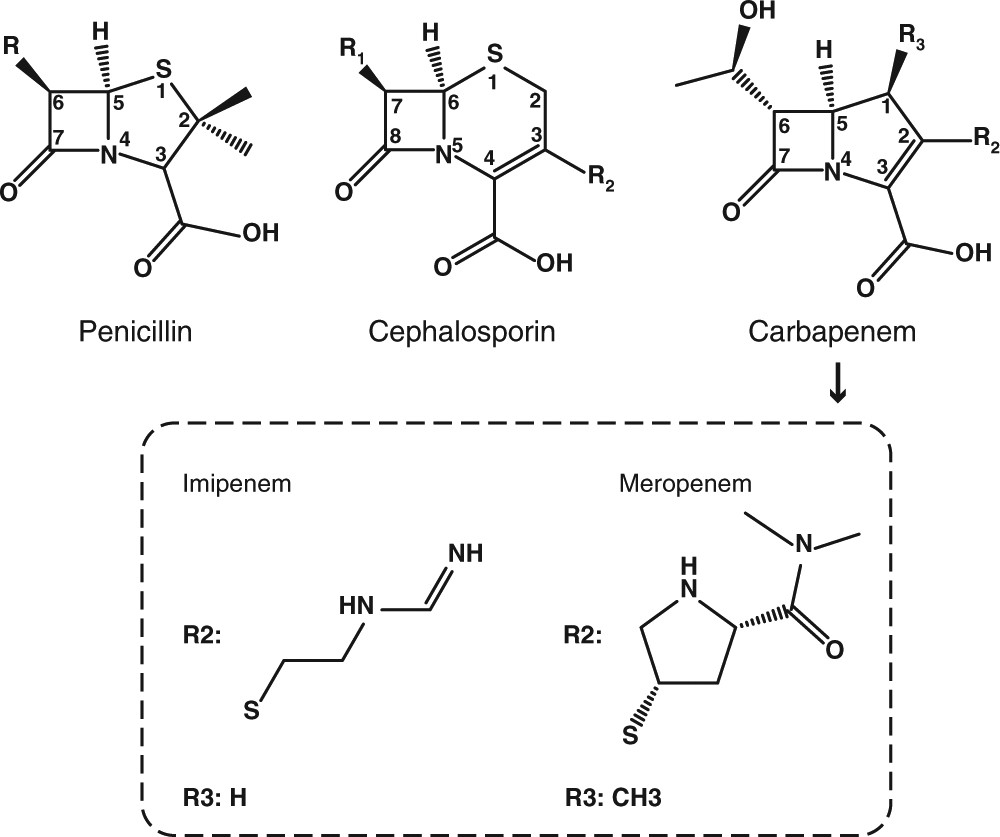
The mechanism of NDM-1-catalyzed carbapenem hydrolysis is distinct from that of penicillin or cephalosporin hydrolysis | Nature Communications

New hydrolysis products of the beta-lactam antibiotic amoxicillin, their pH-dependent formation and search in municipal wastewater. | Semantic Scholar

Crystallographic Snapshots of Class A β-Lactamase Catalysis Reveal Structural Changes That Facilitate β-Lactam Hydrolysis* - Journal of Biological Chemistry

Active-Site Protonation States in an Acyl-Enzyme Intermediate of a Class A β-Lactamase with a Monobactam Substrate | Antimicrobial Agents and Chemotherapy




![betalactam_pharm [TUSOM | Pharmwiki] betalactam_pharm [TUSOM | Pharmwiki]](https://tmedweb.tulane.edu/pharmwiki/lib/exe/fetch.php/betalactams.png?w=600&tok=3379e1)